This page is part of the HL7 Europe Laboratory Report (v0.1.0: STU 1) based on FHIR (HL7® FHIR® Standard) R4. The current version which supersedes this version is 2.0.0. For a full list of available versions, see the Directory of published versions
There are two ways - in principle - for representing a Laboratory report in HL7 FHIR:
In many European countries:
On the other hand, typical HL7 FHIR consumers expect to get Laboratory Reports by searching per DiagnosticReport.
The team looked therefore for a solution balancing the two approaches (FHIR document and DiagnosticReport), taking into account the R5 DiagnosticReport design pattern where the DiagnosticReport Vs Composition relationship is directed from the DiagnosticReport to the Composition resource.
In brief:
The following figure graphically summarizes the described design approach.
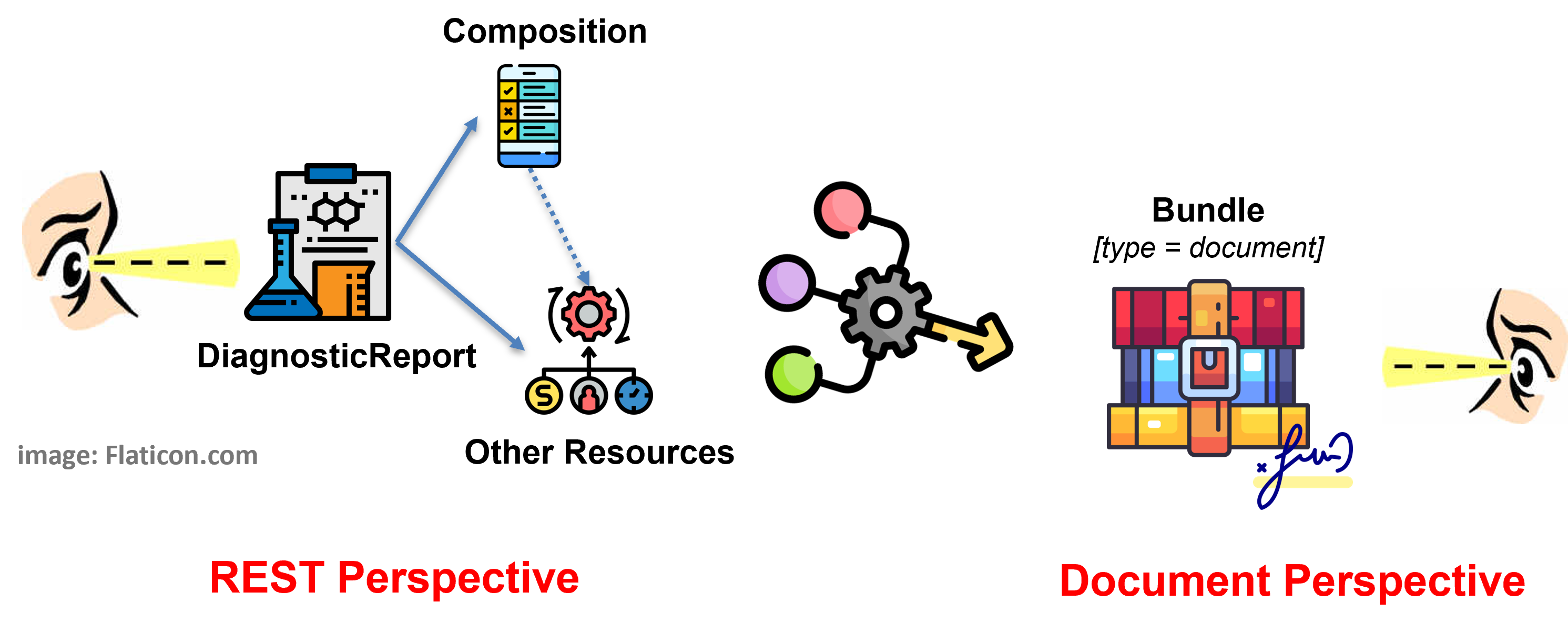
Figure 1 - Overview of the report design approach
The authors are aware of the fact that this choice requires additional work to the creator, requesting to consistently record in both DiagnosticReport and Composition a set of information. However they believe, that it enables for more options for the consumer:
To support the described approach, this guide allows for the pre-adoption of the R5 rules for the inclusion of the resources in a document Bundle, that is:
“The document bundle SHALL include only: <..> The supporting information: Any resources that are part of the graph of resources that reference or are referenced from the composition set, either directly or indirectly (e.g. recursively in a chain)”
in opposition to the R4 rules requiring that, with the exception of the Provenance resource and the Binary including the stylesheet, only resources directly or indirectly - referred from the composition can be included.
This choice is justified by the fact that:
However, this choice it is not imposed, so that usage contexts whishing to keep a full consistency with R4 rules, may used the DiagnosticReportReference extension to refer the DiagnosticReport from the Composition. The adoption of this extension implies the presence of a circular reference Composition to/from DiagnosticReport.